
TruLeaf Medical was founded in 2017 by the late Prof. Uri Rosenschein, Netanel Benichou and Benjamin Spenser, who bring vast experience in structural heart disease, valve design and startup company management.
Our mission is to bring an ultimately user-friendly, Transcatheter Valve Replacement solution that will be available to every cardiologist and valvular regurgitation patient. Our focus is on procedure simplicity, safety and efficacy.
TruLeaf’s approach will bring the solution for the long-lasting problem in an elegant and safe way.
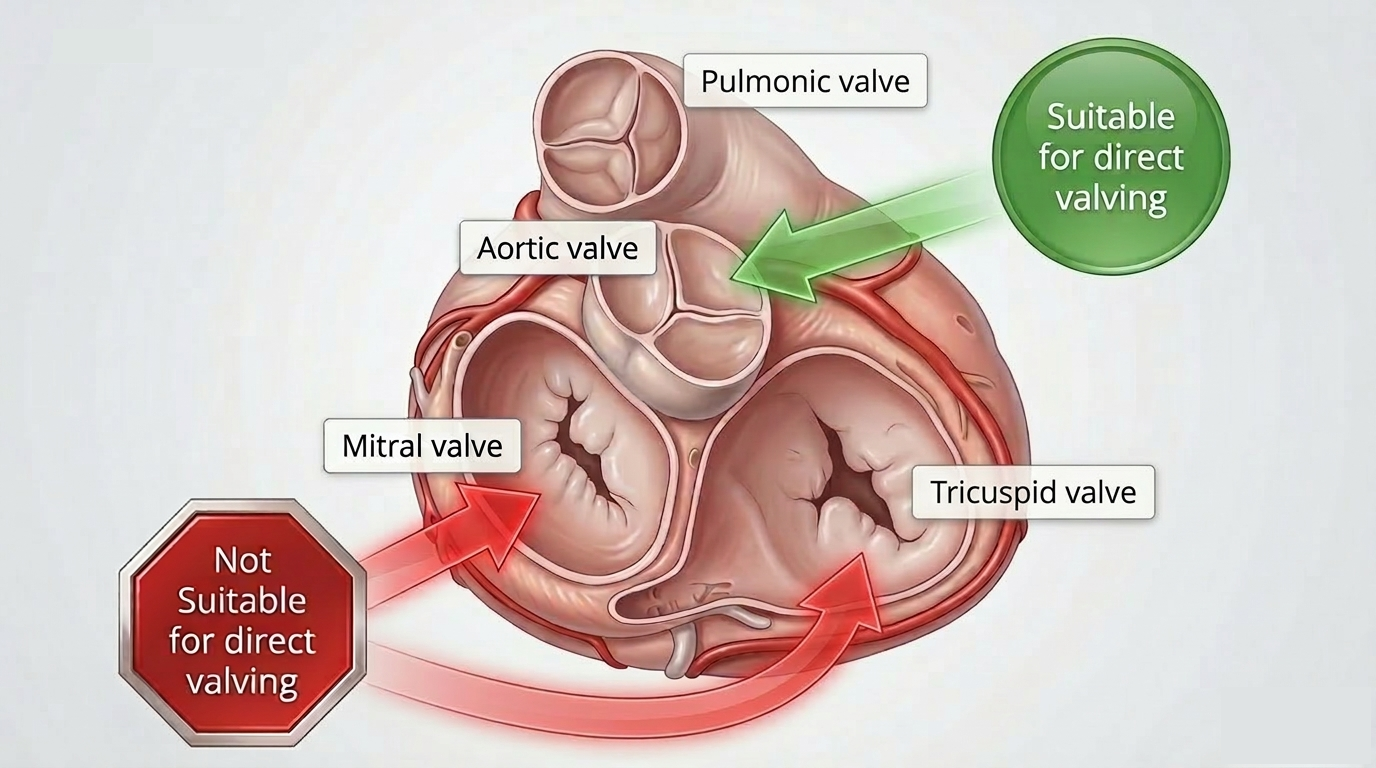
TMVR poses new challenges in comparison with TAVI:
Robust anchoring of the valve to the native annulus
Implantation in a minimally invasive approach – transcatheter
Creation of a simple and safe procedure
Elimination of paravalvular leaks (PVL)
Elimination of Left Ventricular Outflow Tract (LVOT) obstruction
Minimization of disturbance to the cardiac structure
Large valve area and high work blood pressures
Valve prosthesis durability
December 7, 2025
Allmed has completed catheter-based implantation of a valve developed by subsidiary Therolife in two patients with severe tricuspid regurgitation, and continues to recruit patients for trials in additional countries. The two patients, who suffered from advanced heart failure due to severe tricuspid valve leakage, received the treatment as part of “compassionate care” after conventional medical options were not suitable for them. According to company reports, their condition has improved and they are scheduled for discharge in the coming days.
October 9, 2025
TruLeaf Medical, owned by Allmed Solutions, successfully implants its RoseDoc docking device in two patients in India, the first step in a two-stage catheter-based procedure to replace faulty tricuspid and mitral valves without open-heart surgery. The procedure was successfully performed Sept. 5 in India on two patients suffering from severe, treatment-resistant congestive heart failure caused by leaking tricuspid valves. Both patients had no remaining therapeutic options and were treated under a compassionate care pathway.
August 14, 2022
After revolutionizing heart catheterization procedures in the world, the Israeli engineers want to do it again – and this time they are turning to the heart valve market, which is estimated at about 15 billion dollars a year in the US alone. TruLeaf announces it has completed the development of its revolutionary valve prosthesisdasdasdasdasd
May 13, 2020
Allium Medical (TASE: ALMD), an Israeli medical device company, specializes in minimally invasive technologies subsidiary, TruLeaf Medical, has successfully tested a novel approach for transcatheter mitral valve replacement in patients suffering from severe mitral valve regurgitation during a long-term trial on large animals.
April 5, 2020
TruLeaf Medical has received a 5M NIS grant from the Israeli Innovation Authority. This grant follows an additional 2.6M NIS grant received during the month of March 2020.
The grant was approved for the continuation of development of a minimally invasive trans catheter mitral valve replacement. TruLeaf, a daughter company of Allium Medical, is developing a system for the replacement of the mitral valve in patients suffering from severe mitral regurgitation, inserted using catheterization techniques.
The grant will be used for furthering the R&D activities, TruLeaf’s IP and design freeze towards a first clinical study in human patients.